A lot of new information has emerged since the appearance of the original article in 2011. New species and additional distribution data have led to a better understanding of how, where, and why different
Apistogramma species-groups are located where they are. I am certain that additional new data will appear in the future that will modify this, but for now this is how I see how the paleogeography of South America has affected the phylogeny and distribution of
Apistogramma species-groups.
In general, the geographic distribution, or biogeography, of fishes in tropical South America is due to the many hydrologic pathways and barriers. This is particularly true for small fishes. Favorable hydrologic pathways allow easy distribution of fauna. On the other hand, hydrologic barriers produce endemism, or a restriction of a fauna from becoming widely dispersed.
For smaller fishes, like
Apistogramma species, such barriers can include wide,
deep river channels, waterfalls and rapids, and even variations in water chemistry and
temperature.
Schmettkamp (1982) was the first
author to report that most
Apistogramma-species-groups have a regional pattern of
distribution. Of course, this shouldn’t be a surprise. By definition, a monophyletic
species-group arises from a single species and then migrates outward from its original
location. Where a distributional range is disjunct, it is an indication that the
species-group might be either polyphyletic (having several different ancestors) or that
there has been some kind of change in the hydrologic history of the region.
Apistogramma species typically inhabit the
shorelines of small streams, lakes, pools, in flooded forests, and floating meadows. In
these locations they are typically found in shallow water, living among snags, leaf
litter, and aquatic or bog plants. They are almost never found in the open main channel
of major rivers. Their small size prevents them from readily crossing major channels
without some kind of assistance.
Apistogramma species with extensive ranges of over
500 km (300 mi.) are anomalies. Most species previously thought to have large ranges are actually a group of similar looking but genetically distinct species. The distribution patterns of such similar looking populations also tend to be quite linear. Typically these populations are highly adaptable in their habitat requirements and water chemistry. They typically inhabit all types of white-, clear-, or mixed clear/blackwater environments.
Many highly endemic
Apistogramma, on the
other hand, tend to be blackwater species. For blackwater species, endemism is most
likely due to their water chemistry requirements. Blackwater habitats, streams in
particular, are usually not very extensive.
We need to realize that distribution patterns of species-groups are based not only on modern drainage patterns, but also on those that occurred in the past. Major alterations in drainage patterns have occurred in South America over the past several million years. These changes affected
the distribution patterns of the fishes in them. Kullander (1986), for example, noted
the presence of the same species - or very closely related species - in the Amazon
drainage of northern Peru and in the headwaters of the Río Orinoco along the eastern
slopes of the Andes. If one looks at present day hydrogeography of South America, there is no connection
between these two areas. Geological studies, however, indicate that several million years
ago the rivers of the western Amazon Basin actually flowed northward along the foothills
of the Andes and emptied into the Caribbean Sea through Lake Maracaibo. Similar
alterations in the past have affected the distribution patterns of many
Apistogramma species-groups.
The following update is the latest proposed distribution and phylogeny for the
Apistogramma species-groups. Papers on the paleogeography of South American rivers by Lundberg et al. (1998) and in Hoorn & Wesselingh (2010), along with genetic studies on
Apistogramma species by Miller and Schliewen (2005), Ready et al. (2006), and more recently Tougard et al. 2017, da Costa et al. 2019, and Estivals et al. 2020, have modified many of my ideas about the ages and relationships of the various species-groups.
As I have written previously, since this isn't a peer-reviewed scientific paper, I will allow myself much more license in suggesting relationships than would a true taxonomist/systematist! I urge those with questions or criticisms to please write me. Only exchanges of views can improve our knowledge of the
Apistogramma species-groups, their distribution, and phylogenic relationships.
Paleogeography of South America
It should be obvious that evolution of the different
Apistogramma species-groups is strongly linked to the evolution of the drainage patterns that occurred in tropical
South America over at least the past 35 million years, during which time the genus arose and spread
throughout much of tropical South America. The phylogeny of
Apistogramma species-groups is easier to understand when set on the backdrop of the paleogeography of South America.
If we could see the South American
continent of 40 million years ago, it would look nothing like the continent of today.
The direction in the flow of interior rivers was influenced not only by the rising Andes
Mountains and the Guiana and Brazilian Highlands, but also by structural arches and
sedimentary deposition on the ancient interior craton.
Late Eocene Paleogeography (40 Million Years BP)
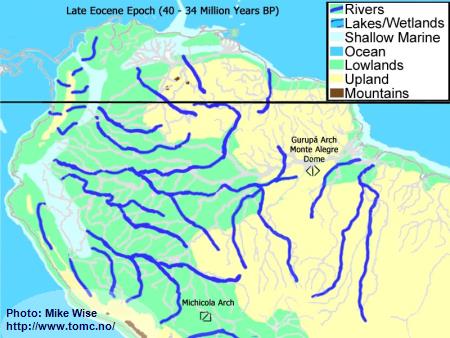
Fig. 1. Late Eocene Paleogeography
If we could see the South American continent as it was during the Late Eocene Epoch 34-40 million years ago it would look very different from what we see today. The Andes Mountains were a chain of islands being pushed up along the western edge of the continent and separated from it by shallow marine seaways. There was no transcontinental Amazon River system. The ancient highlands in the east were connected. The Gurupá Arch and associated Monte Alegre Dome formed a continental divide for the eastern part of South America. Rivers to the east and north drained into the Atlantic Ocean. Rivers to the west flowed into shallow seaways that were separated from the Pacific Ocean by the rising Andean island arc. South of the Michicola Arch, in central Bolivia, rivers flowed south around the cratonic highlands and into the Atlantic Ocean.
Oligocene Paleogeography (34 -23 Million Years BP)
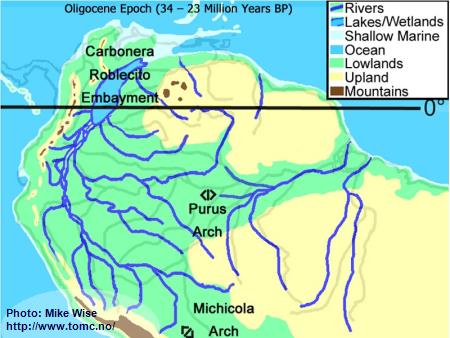
Fig. 2. Oligocene Paleogeography
Prior to 25 million years ago, the Andes Mountains, which started as a volcanic
island arc (similar to present day Japan), finally welded to the South American continent.
To the east, the Guiana and Brazilian Highlands, together with the adjoining Purus Arch
formed highlands on the South American craton. These upland features produced a
hydrological continental divide through the northern half of South America. Rivers to
the west of the divide flowed westward in a sub-Andean River system that followed the
eastern margins of the rising Andes. The system flowed northward into the
Carbonera-Roblecito Embayment in northern Colombia. This embayment was a freshwater
delta much of the time, but occasionally was inundated by marine waters. Rivers draining
the western Guiana Highland flowed westward into the same river system. East of the
Purus Arch, rivers originating in the southern Guiana Highlands and northern Brazilian
Highlands flowed into the proto-Amazon and proto-Tocantins river systems. The Michicola
Arch in central Bolivia appears to have been a highland area that formed a hydrologic
divide between the sub-Andean River system of western Amazonia and the proto-Paraná
system to the south. During much of the period, the proto-Paraná was a freshwater system
that emptied into a shallow marine embayment at the mouth of the present day Río de la
Plata. Several times during the Late Oligocene to Middle Miocene, major inundations by
shallow epicontinental seas (Paranan Sea) filled large parts of the proto-Paraná system.
Early Miocene Paleogeography (23 - 18 Million Years BP)
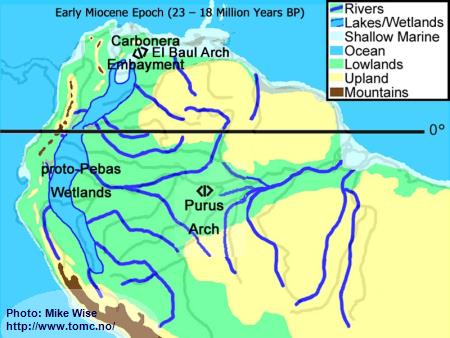
Fig. 3. Early Miocene Paleogeography
Major mountain building in the central and (locally) northern parts of the Andes Range occurred at the beginning of the Miocene Epoch. Uplift of the range caused down-warping of the eastern foreland basins, creating a roughly north-south trough in which the Sub-Andean River flowed. Mountain building in the northern Andes cut off the Sub-Andean River system from its mouth near present day Lake Maricaibo. The waters of the river were forced to flow eastward where the El Baul Arch restricted its flow to the Atlantic. This caused much of the Sub-Andean River basin to be flooded with freshwater. This proto-Pebas Wetland is believed to have been more like an Everglades-like biotope than a large lake. Periodically, northern parts of the system were inundated by marine waters from the Atlantic Ocean. Waters of the lower Amazon basin, east of the Purus Arch, flowed eastward at least as far as the present day Rio Xingu where it emptied into a marine embayment. The proto-Amazon system, originating in the Late Oligocene, included parts of the Rio Solimões east of the Purus arch, the Rio Madeira below the Rio Mamoré, and the Rio Negro below the Rio Branco. The Rio Tocantins system was separated from the Proto-Amazon by a marine embayment.
Middle Miocene Paleogeography (18 - 13 Million Years BP)
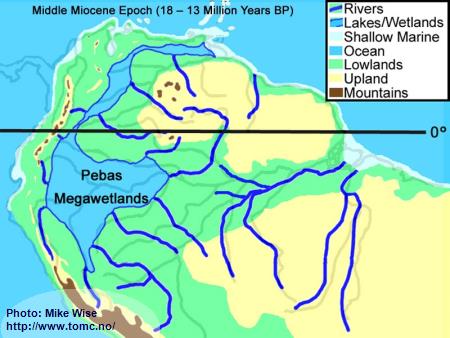
Fig. 4. Middle Miocene Paleogeography
The Middle Miocene Epoch started with continued uplift of the central and northern Andes. With increased height, the mountains trapped more moisture from the Atlantic, forming a wetter climate than is seen in the Amazon Basin today. Rivers flowing eastward out of the Andes meandered back and forth over wide areas, depositing thick layers of sediment at the base of the mountain range. These alluvial fans expanded to form megafans at the base of the range, particularly in area of the present day central and northern Andes foothills. Megafans can be thought of as being similar to river deltas except that they are deposited above water. The megafans buried the western parts of the Sub-Andean River valley, forcing the Sub-Andean River system and wetlands to the east. With increased rainfall, more of western Amazonia, which sat close to sea level, was flooded in freshwater swamps called the Pebas Megawetlands. This is what was previously thought to be an epicontinental lake named Lago Amazonas. Periodically, marine waters invaded the wetlands from northern inlets. Near the end of the Middle Miocene, the El Baul Arch, in present day Venezuela was entirely breached, allowing water to enter the Atlantic near the Orinoco delta. Little change occurred in the proto-Amazon system, east of the Purus Arch.
Late Miocene Paleogeography (13 -5.5 Million Years BP)
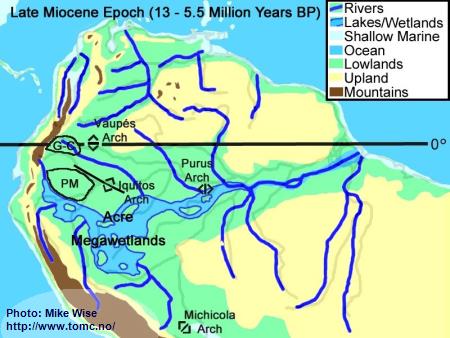
Fig. 5. Late Miocene Paleogeography
PM = Pastaza Megafan; G-C = Guayabero-Caquetá Megafan
The late Miocene Epoch was characterized by increasingly rapid uplift of the central and northern Andes, including the uplift of the Vaupés Arch. The Vaupés Arch blocked northward flow of the southern part of the Sub-Andean River system and initiated the onset of separate Orinoco and Amazon systems. Megafans expanded, becoming regional in size. The rivers on them meandered over wide swaths of land. Two of these megafans appear to have had major effects on the distribution of
Apistogramma species-groups. These are the Pastaza Megafan, which was deposited between the present day Río Putumayo and the Río Tigre drainages, and the Guayabero-Caquetá Megafan, which formed between the Iquitos Arch and the Vaupés Arch. The Guayabero-Caquetá Megafan includes the upper parts of the present day Río Caquetá and Río Guaviaré systems. The southern part of the Sub-Andean River system was blocked to the north by the Vaupés Arch, to the east by the Purus Arch, and to the south by the Michicola Arch. This blockage inundated parts of the central Andean foreland basins, forming the Acre Megawetlands. Eventually, the Purus Arch was breached. Waters from the southern part of the former Sub-Andean River system flowed eastward into the proto-Amazon, forming the present transcontinental Amazon River. Stream piracy increased. In the Rio Negro region, first the Rio Branco and later the Rio Uaupés systems were pirated from the northern (Proto-Orinoco) part of the Sub-Andean River system and incorporated into the present Rio Negro system. The Proto-Rio Madeira pirated much of Sub-Andean system north of the Michicola Arch (Rio Mamore & Rio Beni systems).
Pliocene Paleogeography (5.5 - 1 Million Years BP)
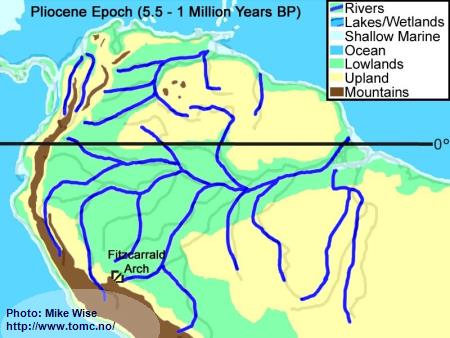
Fig. 6. Pliocene Paleogeography
By the start of the Pliocene Epoch, the river drainages were similar to that of present day South America. The only exception was in the upper drainages of the present day Rio Acre and Rio Madre de Dios, which flowed westward into the Peruvian Amazon. About 4 million years ago, a series of east-west trending submarine volcanoes, resting on the Nazca oceanic tectonic plate west of South America, were subducted below the South American continental plate. This caused a bulge in the continent called the Fitzcarrald Arch. The Fitzcarrald Arch blocked northward flow of the Acre and Madre de Dios systems into the Peruvian Amazon. These two drainages were forced to change their flow to the southeast, into the Rio Purus and Rio Madeira systems. In the Orinoco drainage, continued uplift of the Andes repeatedly filled in the channel of the Orinoco, forcing it eastward as far as the western edge of the Guiana Highland.
Pleistocene Paleogeography (2 million to 10,000 Years BP)
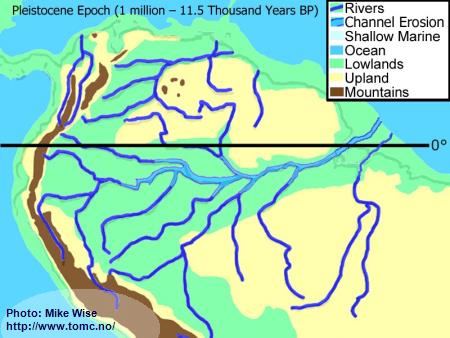
Fig. 7. Pleistocene Paleogeography
The Pleistocene Epoch was influenced more by radical climatic fluctuations than by tectonic processes. The Pleistocene Epoch is known for a series of ice ages in the Northern Hemisphere, yet these had a worldwide affect - even in the tropics. At the beginning of the Pleistocene Epoch, 2 million years ago, most of the rivers in South America were flowing in the same general channels in which they flow today. Sediments eroded from the Andes by the headwaters of the Amazon, Orinoco, and Paraguay had filled in marine embayments at their mouths.
At least four major and an equal number of minor ice ages, with equally spaced warmer interglacial periods, occurred during the Pleistocene Epoch in the Northern Hemisphere. So much water was trapped in continental and mountain glaciers that during glacial periods sea level dropped approximately 100 m (300 ft.) worldwide. Not only did it expose wide areas of previously submerged coastal areas, but it increased the gradients of the rivers. By the end of the Pleistocene, the main channel of the Amazon in many locations flowed 100 m (300 ft.) below the surrounding land. Tributaries cut deeper channels, too, but not as deep. High waterfalls and rapids separated the lower parts of these rivers from their upper parts and isolated the fauna of the northern banks of the Amazon from those of the southern banks.
Although the most severe climatic changes occurred in the Northern Hemisphere, the Southern Hemisphere was affected, too. In South America, during periods of maximum glaciation, many tropical areas cooled. Andean mountain glaciers expanded and trapped precipitation. This led to a drier climate. In many places, the tropical rainforests of the Amazon Basin were replaced by savanna and open montane forests that were more tolerant of the cooler, drier environment. Tropical rainforests retreated to areas where the orographic effects of mountains and highlands still provided enough rainfall to supply their needs.
Holocene Geography (10,000 Years BP-Present)
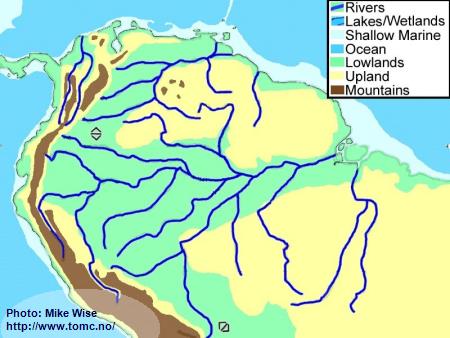
Fig. 8. Holocene Geography
Warmer temperatures, increased rainfall, and a rise in sea level came with the end of the Pleistocene Ice Ages. The climate became warmer and wetter. Increased rainfall helped fill the deep channel of the Amazon with freshwater. Rainforests again invaded the central Amazon Basin.
A Proposed Distribution of the Apistogramma-Species-Groups
The
Apistogramma species-groups listed in this paper are my attempt to break the genus into groupings of species that share physical and behavioral characteristics. For the most part, they also show a common pattern of distribution. Those interested in the descriptions of my species-groups can find them in the online article, “A description of
Apistogramma species-groups” (Wise, 2021). A list of species can be found in the online article, “
Apistogramma Species List By Species-Groups/Complexes August, 2022” (Wise, et al. 2022). Both of these works are based primarily on the works of Koslowski (2002) and Stawikowski (2005), which includes the preliminary genetic findings of Miller & Schliewen (2005). New species and distribution data have been used to modify their works (Tougard et al., 2017, da Costa et al., 2019, Estivals et al., 2020).
The
Apistogramma species-groups used in these articles and the proposed distribution of these species-groups tend to conform closely to the paleohydrology of South America over the past 25 - 30 million years. Altogether, they indicate that the species-groups are for the most part valid – and can be used to predict which groups/complexes can be expected in unexplored regions of South America.
Although we have no direct fossil evidence, the genus
Apistogramma appears to have a rather long history, probably arising from its geophagine ancestors sometime during the late Oligocene or early Miocene Epoch, possibly over 30 million years ago.
Based on locations where more ancestral forms of species-lineages occur, the ancestral forms probably originated somewhere along the northern periphery of the Brazilian Highlands. They probably were casually monogamous, with little sexual dimorphism - similar to species in the
Apistogramma-
regani-lineage. This area seems to be the source of ancestors for the
Apistogramma-
pertensis- and
Apistogramma-
trifasciata-lineages, too, since primitive examples of these lineages also occur in the region today. The ancestors of these lineages eventually radiated outward in all directions.
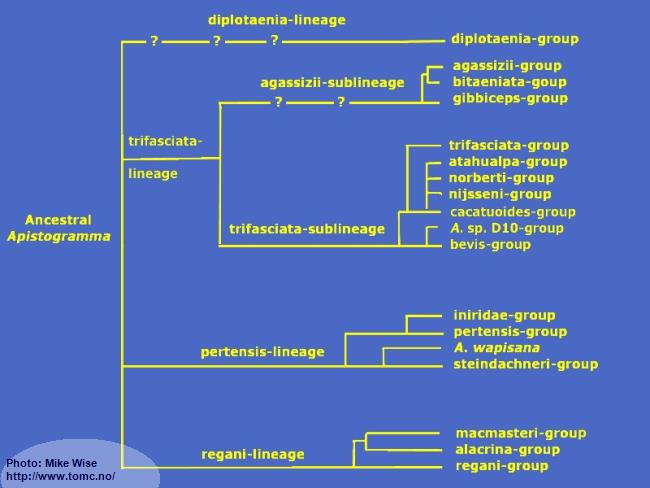
Fig. 9. A Proposed Phylogeny of Apistogramma Species-Groups
Apistogramma-regani-Lineage
The
Apistogramma-
regani-lineage is widely distributed through most of tropical South America east of the Andes Mountains. Ancestral species of the
Apistogramma-
regani-lineage appear to have established themselves along the northern margins of the Brazilian Highlands very early. From there, species of the
Apistogramma-
regani-group migrated first north, then west and south to inhabit waters of the Amazon and Paraguay systems. The
Apistogramma-
regani-group became the source for species of the
Apistogramma-
alacrina- and
Apistogramma-
macmasteri-groups in the Orinoco system.

Fig. 10. A proposed distribution for the regani-lineage
Apistogramma-regani-Group
Early migration was northeastward into the eastern part of the proto-Amazon system, where the kullanderi-, Xingu- and caetei-complexes arose. Other species-complexes moved south and west.
Eastern Amazon Basin
Early migration was northeastward into the eastern part of the proto-Amazon system, where the kullanderi-, Xingu- and caetei-complexes arose. Other species-complexes moved south and west.
Species of the eastern Amazon Basin include three species-complexes that originated in the northeastern part of the Brazilian Highlands, the kullanderi-, Xingu-, and caetei-complexes. They were originally restricted to the proto-Amazon and proto-Tocantins by a marine embayment to the north and the Gurupa Arch/Xingu Dome to the west. Later in its history the Eastern Amazon Basin was invaded by species-complexes from the Central Amazon Basin.
Apistogramma-kullanderi-Complex
I consider the kullanderi-complex the most primitive species-complexes in the regani-group, and one of the more primitive within the genus. Species exhibit features very similar to some species in the closely related genus Gymnogeophagus, which are mostly found on the southern side of the Brazilian Highlands. I believe it arose sometime during the Oligocene Epoch.
Species of the kullanderi-complex are restricted to rivers in the upper Rio Xingu and Tapajós systems (Fig. 11).
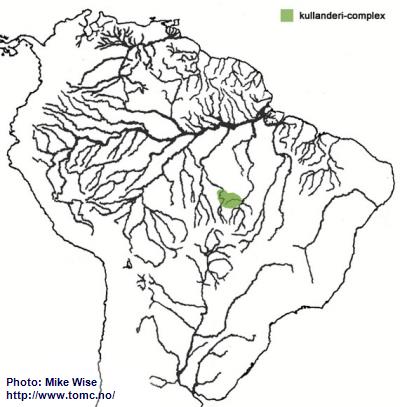
Fig. 11. Distribution of the kullanderi-complex
Apistogramma-sp.-"Xingu"-Complex
The species of the Xingu-complex show many features in common with those of the kullanderi-complex. It is likely that they arose from an ancestral kullanderi-complex species in the northern Brazilian Highlands sometime during the early Miocene. Like species of the kullander-complex, the Xingu-complex arose in the upper reaches of the Xingu and Tapajós systems, where species of the Xingu-complex still occur (Fig. 12.). From there, species of the Xingu-complex migrated down the Rio Xingu. There is also a species (
A. sp. “Jabuti”) that is found in the lower Tapajós. No species of the complex are known to occur in the middle reaches of the river, however. This could be due to insufficient collection. On the other hand, it is also possible the Xingu-complex migrated into the lower Tapajós from the lower Xingu.
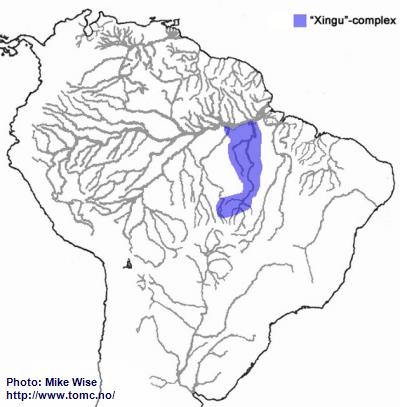
Fig. 12. Distribution of the "Xingu"-complex
Apistogramma-caetei-Complex
Species of the caetei-complex probably originated from a species of the Xingu-complex somewhere the northern Brazilian Highlands. From there it migrated down the Rio Araguaia and into the lower Rio Tocantins. This migration probably occurred very early in the history of the genus, sometime during the early Miocene. First a marine embayment at the mouth of the proto-Amazon and later the broad main channel of the Amazon prevented the caetei-complex from migrating to the north side of the Amazon. Lowering of sea level during glacial periods of the Pleistocene opened new areas of the Atlantic coastal plain, allowing the caetei-complex to invade the Ilha de Marajó and areas north of the Serra de Tiracambu uplands (Fig. 13).

Fig. 13. Distribution of the caetei-complex
Central Amazon and Paraguay Basins
Species of three regani-group species-complexes occur in the central Amazon basin, primarily the Rio Madeira System. Two of these, the resticulosa-complex and commbrae-complex are closely related. The third, less related complex, is the regani-complex. The Paraguay Basin is dominated by the borellii-complex, although resticulosa- and commbrae-complex species are found there. The regani-complex shows a more convoluted distribution.
Apistogramma-resticulosa-Complex
The ancestor of the resticulosa-complex probably originated in the western Brazilian Highlands (Fig. 14). From there, species of the complex migrated to both the southwest and southeast along the southern part of the Sub-Andean River system. This probably occurred early in the evolution of the genus, at least by the middle Miocene. Species of the complex reached southeastern Peru by the start of the Pliocene, but further migration along the Sub-Andean river system was blocked by the rise of the Fitzcarrald Arch. Other species of the complex started migrating southeastward into the upper reaches of the present day Rio Mamoré and lower Rio Guaporé, then a part of the Sub-Andean river system. During the late Miocene or Pliocene, these eastern rivers of the Sub-Andean river system were pirated by the lower present day Rio Madeira. Species of the complex were now able to migrate down the Madeira and along the southern banks of the Amazon where they inhabit the lower reaches of its southern tributaries. It was only able to migrate into a limited number of northern tributaries of the Amazon located downstream of the Rio Negro. One questionable species of the complex (
A. pleurotaenia) occurs in the Río Paraguay. It probably arrived during the Pleistocene, together with species of the commbrae-complex and trifasciata-group, when the uppermost part of the Río Guaporé was pirated by the Río Paraguay.
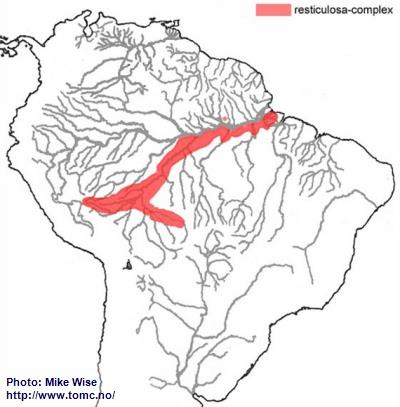
Fig. 14. Distribution of the resticulosa-complex
Apistogramma-commbrae-Complex
The ancestor of the commbrae-complex appears to have been a species of the resticulosa-complex that lived in southern part of the Sub-Andean River system. From there, species of the commbrae-complex migrated east and west (Fig. 15). The rise of the Fitzcarrald Arch during the Pliocene prevented further westward migration. At this time the commbrae-complex was restricted to the upper part of the present day Madeira (Beni, Mamoré, and Guaporé). Eventually, part of the uppermost Madeira (Guaporé) was pirated by the Río Paraguay. This probably occurred during the Pleistocene, when lower sea levels increased stream gradients and increased downward cutting of stream channels. Once the uppermost Guaporé became part of the Paraguay, species of the commbrae-complex eventually migrated into the lower Río Paraguay and Uruguay systems.
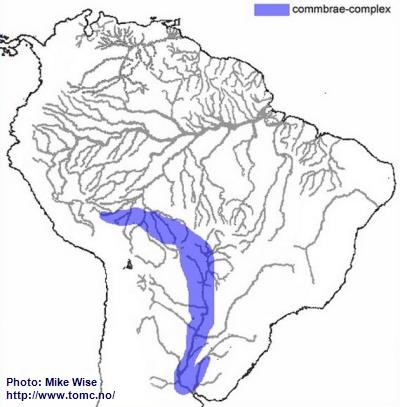
Fig. 15. Distribution of the commbrae-complex
Apistogramma-borellii-Complex
Apistogramma borellii (sensu lato) is widely distributed down the main channel of the Río Paraguay system of Brazil, Bolivia, Paraguay, and northernmost Argentina (Fig. 16). Koslowski (2002) wrote that
A. borellii had more features in common with species of the macmasteri-group than the regani-group. Miller and Schliewen (2005), however, could not find any close relationship between the two groups.
I believe that
A. borellii arose from a resticulosa-complex ancestor that had found its way into the Paraguay. Similarities with species of the macmasteri-group might be explained by species convergence. Both groups live in more open savanna/plains environments. Each group would select similar features to best adapt to similar environments. It is very recent in origin, probably during the Pleistocene. Although
A. borellii is presently considered to be a single species, its extensive distribution is unusual for a species of the genus. It is very likely that the many isolated populations of the species will eventually be genetically recognized as many different species.
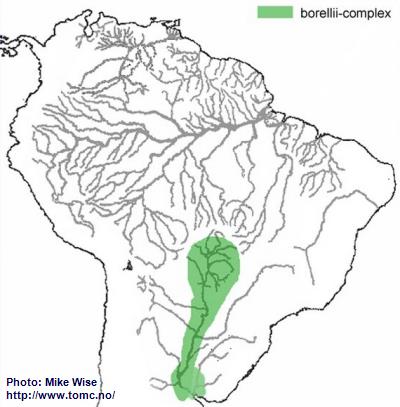
Fig. 16. Distribution of the borellii-complex
The Genus Apistogrammoides
The genus
Apistogrammoides presently consists of only one species. Just like
A. borellii it is very likely that the many isolated populations of the species will eventually be genetically recognized as several different species. The genus is distributed from the upper Ucayali downstream as far as the Colombian Amazon (Fig. 17). This species probably arose from a resticulosa-complex species that became separated from other resticulosa-complex species by the uplift of the Fitzcarrald Arch during the Pliocene. In isolation, it developed into a separate genus and has migrated downstream as far as the Colombian Amazon.
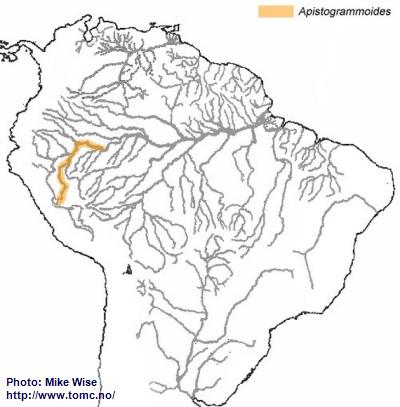
Fig. 17. Distribution of Apistogrammoides
Apistogramma-regani-Complex
When looking at the distribution of the regani-complex (Fig. 18) it appears very disjunct. If only looking at the present hydrology of South America it might appear that the regani-complex is actually polyphyletic, originating from two or more different ancestral species. But when I look at the past hydrology of South America I recognize a monophyletic origin for the complex with migration that moved in two directions at two different time.
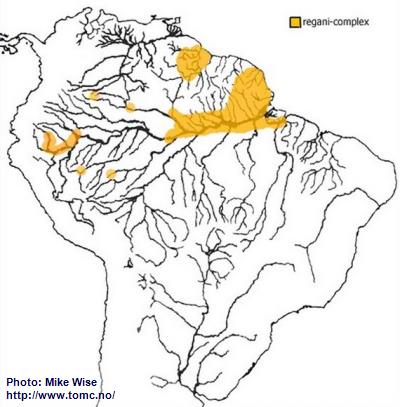
Fig. 18. Distribution of the regani-complex
The regani-complex species seem to have originated somewhere along the northwestern flanks of the Brazilian Highlands (Fig. 19), where there are still several unusual species with a mixture of features of both regani- and eunotus-complexes that still live in this region.
One of its ancestors appears to have moved westward together with other species-groups/complexes along the southern section of the Sub-Andean River system. This certainly occurred before the rise of the Fitzcarrald Arch during the Pliocene, probably in the early to middle Miocene, since species of the regani-complex occur on both sides of this divide. The complex was able to migrate northward along the eastern flanks of the Pebas Megawetlands. During the middle Miocene it was able to migrate throughout the proto-Vaupés as far as the proto-Branco, all of which was then part of the Sub-Andean River system.
When the Purus Arch was breached during the late Miocene, parts of the Sub-Andean River system were pirated into the newly formed Amazon River. These included the upper Rio Nego Negro, including the Rio Uaupés/Vaupés and Rio Branco, and the upper Rio Madeira system, including the Rio Mamoré, Río Beni, and Río Madre de Dios.
In the west, the regani-complex followed the newly formed Rio Negro to its mouth on the Amazon. In the south, the pirating of southern parts of the Sub-Andean River system by the Madeira allowed regani-complex species to migrate down the Madeira and eastward to the mouth of the Amazon.
Sometime during the Pliocene or Pleistocene the regani-complex was able to migrate over the Branco-Rupununi divide, and enter Atlantic coastal rivers in Guyana, Surinam, and eastern Venezuela during the late Miocene During the Pleistocene, with lower sea levels exposing more land, the complex migrated into the Amapá region of northeastern Brazil
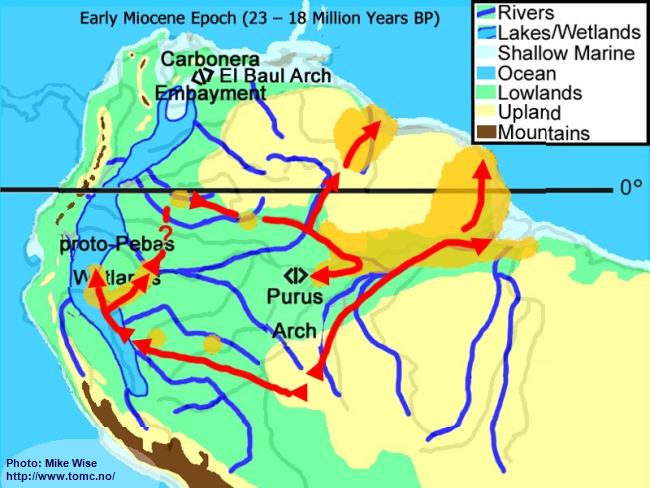
Fig. 19. A proposed migration of the regani-complex
Apistogramma-sp.-"Winkelfleck/Angle-patch"-Complex
The Winkelfleck-complex appears to be closely related to the regani-complex. It probably arose from a regani-complex species somewhere along the eastern edge of the Pastaza Megafan, south of the main channel of the Peruvian Amazon. This probably occurred sometime after the middle Miocene. (Fig. 20).
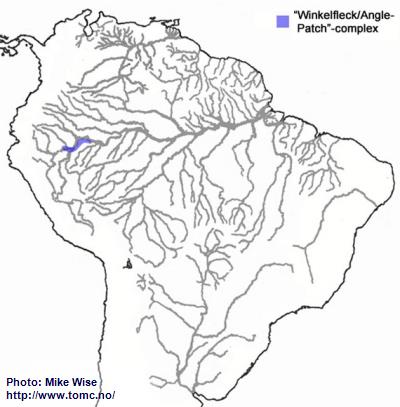
Fig. 20. Distribution of the Winkelfleck/Angle-patch-complex
Apistogramma-eunotus-Complex
The eunotus-complex, like the regani-complex, appears to have originated from species living along the northwestern flanks of the Brazilian Highlands, where eunotus-like species still exist. It also followed the same arcuate course of the Sub-Andean River system westward and then north into western South America, at least as far as the Vaupés Arch (Fig. 21). This migration probably occurred during the middle Miocene, prior to when the Pastaza Megafan started to form.
Although species of the eunotus-complex cannot be subdivided genetically, it can be subdivided by physical features and distribution patterns. Species of the cruzi-subcomplex occur in areas between the northern edge of the Pastaza Megafan and southern flank of the Guayabaro/Caquetá Megafan at elevations above 500m (1600 ft.). This would indicate that these typically lowland species inhabited the area prior to the continued uplift of the Andes and development of the megafans. It appears that the ortegai-subcomplex is a relatively recent offshoot of the cruzi-subcomplex that is derived from an ancestor that lived in the lower Río Napo. The eunotus-subcomplex appears to have originated along the southern and eastern edges of the Pastaza Megafan.
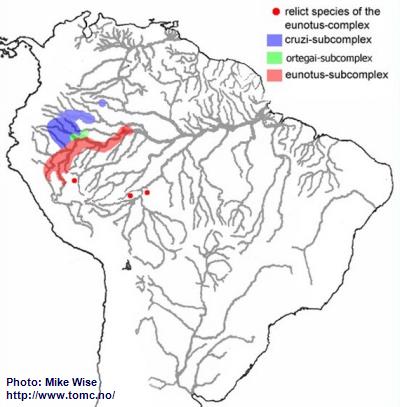
Fig. 21. Distribution of the eunotus-complex
Apistogramma-alacrina-Group
Based on physical characteristics, species of the alacrina-group appear closely related to cruzi-subcomplex species of the eunotus-complex. Species of the alacrina-group and cruzi-subcomplex are both found between northern margin of the Pastaza Megafan and on the Guayabaro/Caquetá Megafan (Fig. 22). The alacrina-group probably arose during the late Miocene after the Purus Arch was breached and the modern Amazon formed. The area north of Pastaza Megafan and Iquitos Arch - which now includes the Caquetá/Japura system, a northwestern outlier of the Amazon basin - became an isolated region of the Peruvian Amazon (Fig. 22). More recently species of the group migrated into nearby headwaters of the Río Guaviare, part of the Orinoco, and the Río Vaupés, part of the Rio Negro.
The alacrina-group can be subdivided into two species-complexes primarily based on their physical form. Species of the alacrina-subcomplex are deeper bodied/higher backed and inhabit waters that are slow moving. Species of the Mitu-subcomplex have more slender bodies and inhabit areas with faster flowing water.
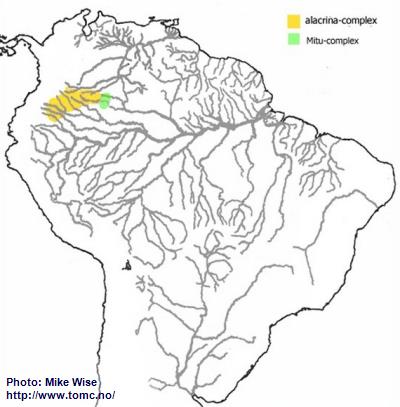
Fig. 22. Distribution of the alacrina-group
Apistogramma-macmasteri-Group
Physically, species of the macmasteri-group appear to be quite similar to species of the eunotus-complex. It probably arose sometime during the Middle to Late Miocene, before the formation of the modern Amazon. Once the Purus Arch was breached and the modern Amazon formed, the northern part of the Sub-Andean River system - along with its fish fauna - became isolated from the southern part. North of the Caquetá/Japura system, an eunotus-complex species was probably cut off from species in the Amazon. In isolation, it gave rise to species of the macmasteri-group.
The species of the macmasteri-group can be split into two species-complexes, the macmasteri- and hongsloi-complex. The macmasteri-complex is further split into two species-subcomplexes, the macmasteri- and hoignei-subcomplex. For the most part, species of the macmasteri-subcomplex are distributed in the upper reaches of the Orinoco system. Those of the hoignei-subcomplex are, for the most part, distributed in left bank streams closer to the main channel of the Orinoco. Species of the hongsloi-complex also occur mostly along the main channel of the Orinoco, but are found in streams on both sides of main channel (Fig. 23).
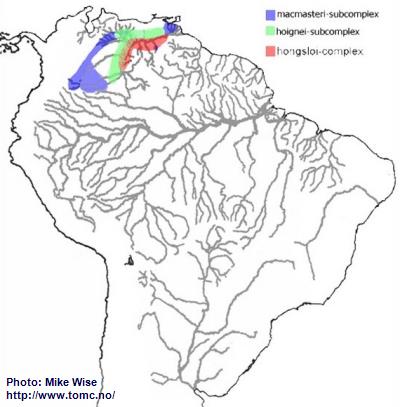
Fig. 23. Distribution of the macmasteri-group
Apistogramma-macmasteri-Complex
The macmasteri-complex species probably originated during the Late Miocene, north of the Vaupés Arch (Fig. 24). Many species of the macmasteri-subcomplex still occur in this area. From this locus, the macmasteri-complex followed the western shoreline of the northern Sub-Andean River system. From there, the species of the macmasteri-complex migrated around the northern edge of the Guiana Highland and into the newly formed Orinoco delta. Development of megafans in northern South America during the Late Miocene pushed the main channel of the northern Sub-Andean River/Orinoco eastward. Species of the hoignei-subcomplex developed primarily west of the channel.
Apistogramma-hongsloi-Complex
The hongsloi-complex probably arose from a macmasteri-complex species after the main channel of the early Orinoco was forced eastward by sediments from the Andes filling in its original western channel. The hongsloi-complex forms tend to occur along both sides of the main channel of the modern Orinoco, along the western flanks of the Guiana Highlands (Fig. 24). It is possible that the hongsloi-complex originated along the southern shoreline of a large llanos lake that existed in the Middle Miocene, prior to a second breaching of the rejuvenated El Baul Arch during the Late Miocene. It would explain its distribution in mostly llanos regions.
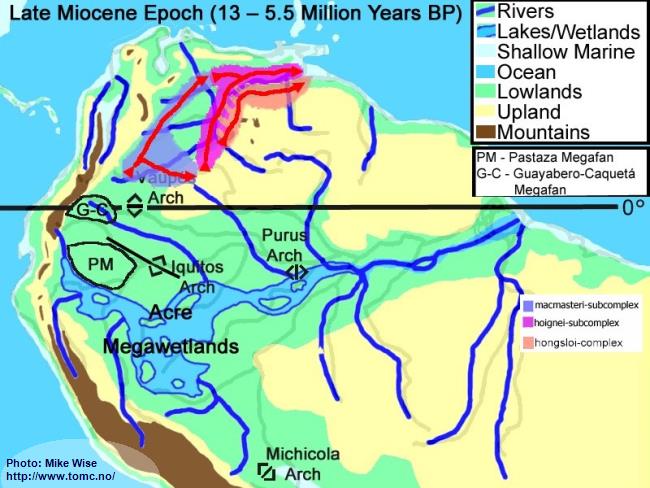
Fig. 24. Migration of the macmasteri-group
Apistogramma-pertensis-Lineage
The pertensis lineage (Fig. 25) represents a genetically monophyletic clade. With a few exceptions, all of the species in the lineage exhibit a complete set of pleisiomorphic cephalic pores. This includes 4 infraorbital pores and 5 dental pores. The exception is the iniridae-group and some individual specimens of the velifera-complex that only have 3 infraorital pores. All species show a prominent spot on Bar 3 on or just above the lateral band. This separates, visually, species of the pertensis-lineage from those of the regani-lineage, which do not possess a lateral spot.
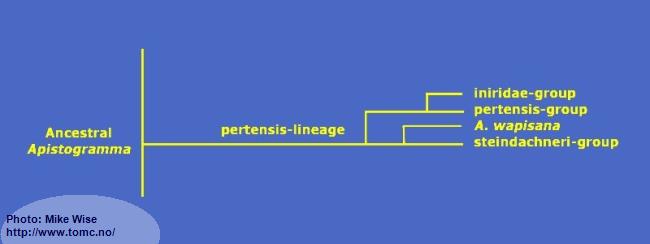
Fig. 25. Proposed phylogeny of the pertensis-lineage
If one looks at the present distribution pattern for the pertensis-lineage it appears that nearly all members are found north of the main channel of the Amazon River (Fig. 26). But the ancestors of the pertensis-lineage actually appear to have originated in the northwestern part of the Brazilian Highlands, where a relict species of the steindachneri-group still occurs. Species of the lineage migrated down the proto-Madeira and into the headwaters of the proto-Amazon, east of the Purus Arch. There, it split into several different species-groups. This probably occurred during the Middle Miocene, prior to the development of the present transcontinental Amazon system that formed in the Late Miocene. Over time it has migrated as far as the middle Rio Orinoco.
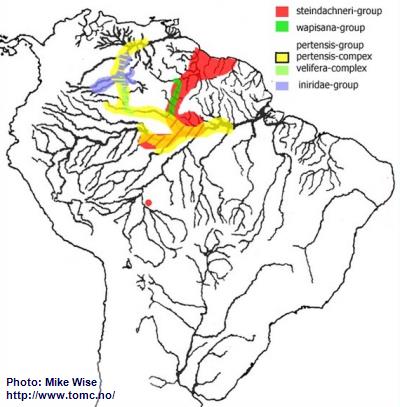
Fig. 26. Distribution of the pertensis-lineage
Apistogramma-steindachneri-Group
The ancestor of the steindachneri-group originated along the northwestern edge of the Brazilian Highlands (Fig. 27). A primitive representative (
A. sp. Rio Preto) still exists in the area.
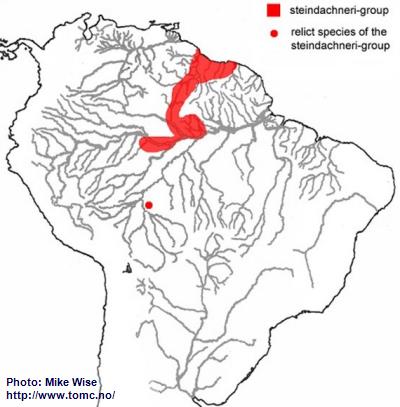
Fig. 27. Distribution of the steindachneri-group
Sometime in the Early to Middle Miocene members of the group migrated down the proto-Madeira and into the upper reaches of the proto-Amazon east of the Purus Arch (Fig. 28). Its distribution included the proto-Negro, which at the time had its headwaters below the Rio Branco. The present upper Negro, the Rio Branco and farther upstream, here called the proto-Uaupés/Branco, was then an eastern tributary of the Sub-Andean River system and flowed to the west. Sometime during the Late Miocene the Rio Branco system was pirated from the Sub-Andean River system by the proto-Rio Negro. Species of the steindachneri-group migrated upstream and into the Rio Branco. Species of the group then crossed over the Branco-Rupununi divide into Atlantic coastal rivers between eastern Venezuela and Surinam. This probably occurred together with species of the regani- and agassizii-complexes during the Pliocene.
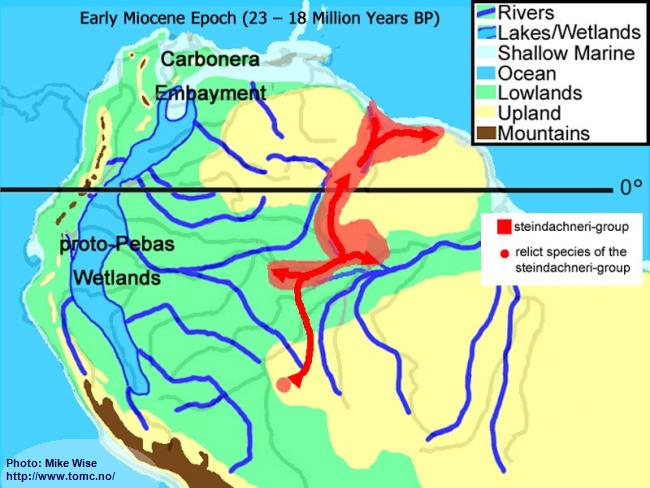
Fig. 28. Migration of the steindachneri-group
Apistogramma-wapisana-Group
According to Miller and Schliewen (2005),
A. wapisana appears to represent a pertensis-lineage species more closely related to the steindachneri-group than the pertensis-group. It certainly exhibits attributes of both of theses species-groups. It is also found in the middle of the present ranges of these two species-groups. It might be an ancestral remnant of an original pertensis-lineage form, but more likely is an offshoot of the steindachneri-group that originated in the upper proto-Negro. Once the proto-Negro pirated the Rio Branco from the Sub-Andean River system during the late Miocene,
A. wapisana was able to migrate into the upper Rio Branco. It must have arrived in the upper Branco after the closure of the Branco-Rupununi divide because it was not able to enter the Guianas. It now inhabits the present day middle Rio Negro and Rio Branco (Fig. 29). A form that may or may not be
A. wapisana has recently been exported from Brazil. For this reason
A. wapisana is labeled here as a species-group.
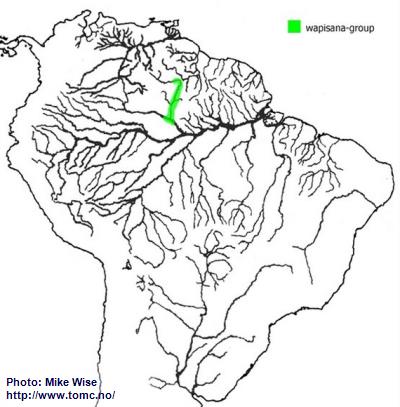
Fig. 29. Distribution of A. wapisana
Apistogramma-pertensis-Group
It is possible that the ancestor of the pertensis-group arose along the western edge of the Brazilian Highlands at about the same time as the steindachneri-group, but it is more likely to have occurred elsewhere and at a later time. Although there are no known species that occur in the upper Madeira, an undescribed species (
A. sp. “Erdfresser/Earth-eater”) is found in the Rio Purus. The pertensis-group produced two different species-complexes – the older and more widespread pertensis-complex and the more recent and endemic velifera-complex.
It is uncertain where the ancestor of the pertensis-complex originated, but the group spread widely throughout the proto-Amazon. This probably occurred during the early or middle Miocene, prior to the breaching of the Purus Arch and development of the present transcontinental Amazon during the late Miocene. Once the present upper Rio Negro was pirated from the Sub-Andean River system, sometime in the late Miocene, the pertensis-group migrated into the upper Rio Negro and eventually into the Orinoco (Fig. 30). For some reason, the pertensis-group never spread up the Rio Branco.
The two species of the velifera-complex appear to be endemic species, specialized to extreme blackwater biotopes. They are found only in the headwaters of the present day upper Rio Negro/Orinoco where they probably originated from a pertensis-complex species (Fig. 30). The velifera-complex possibly is the ancestor of the iniridae-group.
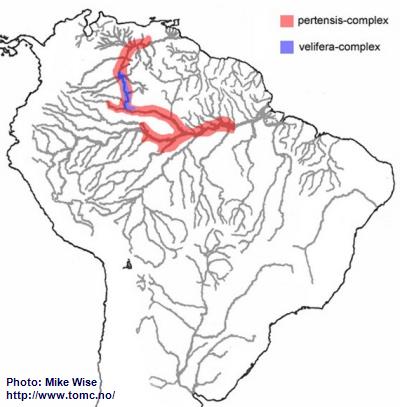
Fig. 30. Distribution of the pertensis-group
Apistogramma-iniridae-Group
The species of the iniridae-group probably arose from a species of the velifera-complex. Like the velifera-complex species, members of the iniridae-group are restricted to blackwater biotopes, but occur only in the upper Río Orinoco drainage (Fig. 31).
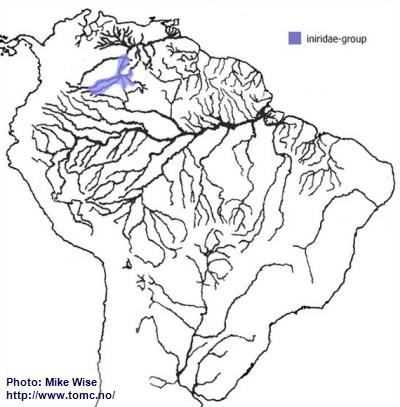
Fig. 31. Distribution of the iniridae-group
Apistogramma-trifasciata-Lineage
The species-groups in the trifasciata-lineage represent a monophyletic clade (Fig. 32) that exhibit a reduced number of cephalic pores when compared to species of the other lineages. It can be split into two sublineages based mainly on the number of dental pores on the head. The species-groups of the trifasciata-sublineage show more plesiomorphic (ancestral) features than do the species-groups in the agassizii-sublineage. There is little doubt that the trifasciata-sublineage is the older and that the agassizii-sublineage arose from it at a later time.
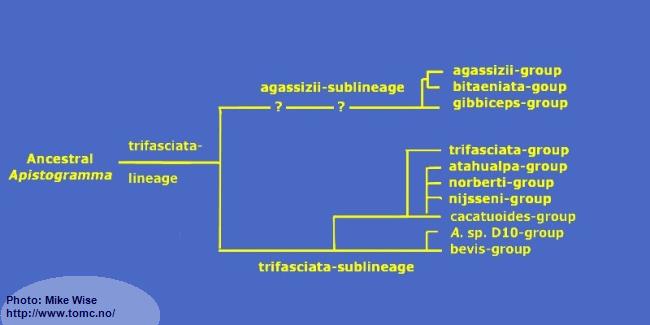
Fig. 32. Proposed phylogeny of the trifasciata-lineage
Apistogramma-trifasciata-Sublineage
It appears that the ancestors of the trifasciata-sublineage originated in the northwestern Brazilian Highlands, since most species-groups are found along the Sub-Andean River system in the southern and western Amazon Basin. All species-groups except two occur in southern and western Amazonia. The brevis-group and its offshoot, the D10-group, can be considered northern outliers.
Apistogramma-brevis-Group
Based on both physical features and distribution, the brevis-group is the most difficult to define of all of the species-groups in the trifasciata-sublineage. Its range is far from that of any other species-group in the sublineage. Presently, the species of the brevis-group are found only in blackwater rivers of the upper Rio Negro/Orinoco region (Fig. 33). They appear most closely related to the cacatuoides-group, which they resemble closely. The brevis-group probably originated from an early cacatuoides-like species. It appears that ancestors of the brevis-group were able to cross into the paleo-Madeira and enter the upper reaches of the proto-Amazon during the Early to Middle Miocene. The species
A. arua,
A. salpinction, and
A. sp. “Doppelfleck/Double-spot” may represent relict forms of this migration. From there it migrated into the proto-Negro. During the Early and Middle Miocene, the proto-Negro ended below the present day Rio Branco. Migration into the upper Negro/Orinoco probably occurred together with that of the pertensis-group, no earlier than the Late Miocene or Pliocene, after the proto-Uaupés-Branco was pirated by the Negro from the Sub Andean River system.
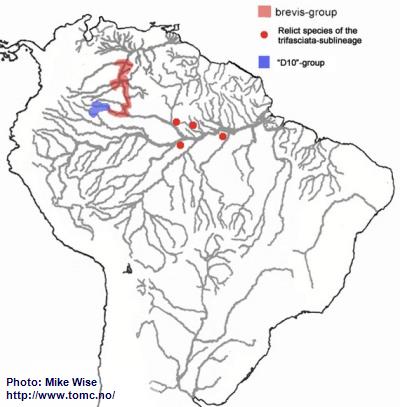
Fig. 33. Distribution of A.-brevis-/"D10"-groups
Apistogramma-sp.-"D10"-Group
Based on its pattern of distribution, the D10-group appears to be an offshoot of the brevis-group (Fig. 33). Although its species in many ways resemble the species in the macmasteri-group, it is a part of the trifasciata-lineage. Species of the D10-group typically possess the trifasciata-lineage’s lateral spot, which species of the regani-lineage lack. Their apparent similarity to the macmasteri-group probably is due to evolutionary convergence. Both species-groups occur in the same region. The D10-group most likely diverged from the brevis-group sometime after the Late Miocene.
Apistogramma-cacatuoides-Group
The ancestor of the cacatuoides-groups appears to have originated in the southwestern Brazilian Highlands sometime in the Early Miocene. During the later parts of the Miocene it first migrated into streams in the upper reaches of the southern Sub-Andean River system, the present day Río Mamoré/Guaporé system (Fig. 34). It then followed this river system into western Amazonia as far as the Pastaza Megafan. This probably occurred early in the Late Miocene. Further migration was blocked by the Iquitos Arch and/or the Guayabero-Caquetá Megafan to the north and the Pebas Megawetlands to the east. With the breaching of the Purus Arch during the Late Miocene, forming the transcontinental Amazon River, the cacatuoides-group was able to expand eastward into the Rio Solimões.
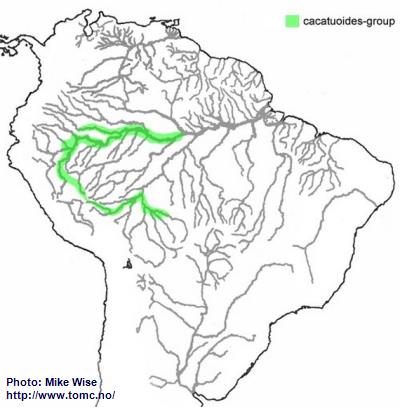
Fig. 34. Distribution of the cacatuoides-group
Apistogramma-nijsseni-Group
The ancestors of the nijsseni-group appear, like other trifasciata-lineage species-groups in western Amazonia to be an offshoot of the cacatuoides-group. Species of the nijsseni-group mostly occur in rivers that now cross the region formed by the Pastaza Megafan (Fig. 35). Species of this species-group are presently known to occur in Ecuador, together with cruzi-subcomplex species, at elevations above 500m (1600 ft.). The only way in which this could occur is by the fish existing in the area before its uplift after Late Miocene. It appears that the nijsseni-group was able to migrate farther north as far as the rise of the Vaupés Arch, which separated the northern part of the Sub-Andean River system from the southern part and blocked further migration. Members of the nijsseni-group tend to be species that are endemic to blackwater and mixed clear-/blackwater biotopes with rather restricted ranges.
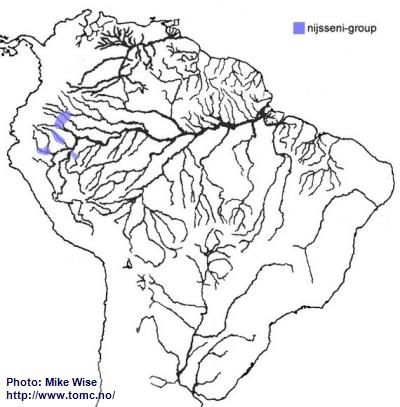
Fig. 35. Distribution of the nijsseni-group
Apistogramma-atahualpa-Group
According to Miller and Schliewen (2005) the species of the atahualpa-group are an early offshoot of the cacatuoides-group. They probably originated from a cacatuoides-group species somewhere along the outer edge of the Pastaza Megafan, probably sometime during the Late Miocene. They are primarily blackwater species and are restricted to such biotopes. The atahualpa-group can be split into two different complexes by their breeding behavior and some minor physical differences, but not genetically. The older atahualpa-complex species, found south of the main channel of the Amazon (Fig. 36), breed in the manner typical for Apistogramma. The barlowi-complex species of the atahualpa-group appear to have undergone fairly recent modification to aid in a mouthbrooding style of reproduction. Like the atahualpa-complex, the species of the barlowi-complex are adapted for and endemic to blackwater tributaries. They, however, are presently known only north of the main channel of the Peruvian Amazon, with one species finding its way into the lower Rio Solimões near Manaus, Brazil (Fig. 36).
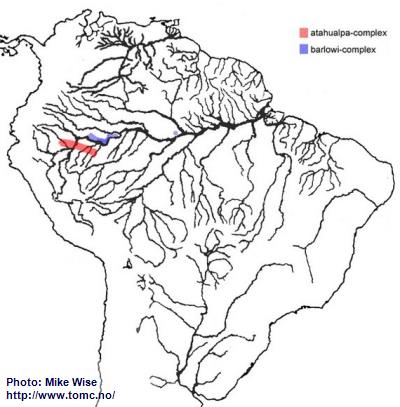
Fig. 36. Distribution of the atahualpa-group
Apistogramma-norberti-Group
The two species presently belonging to the norberti-group appear to be more recent offshoots of the cacatuoides-group, possibly post-Late Miocene.
Apistogramma norberti is found in streams entering the lower Río Ucayali.
Apistogramma cf.
norberti (Lyretail) occurs in the streams around Leticia, Colombia (Fig. 37). The norberti-group probably arose from its cacatuoides-group ancestor on the Pastaza Megafan and then migrated along the upper Amazon.
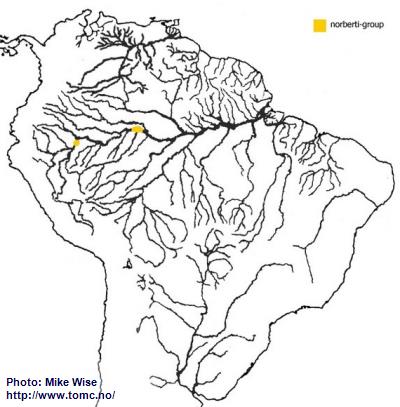
Fig. 37. Distribution of the norberti-group
Apistogramma-trifasciata-Group
The trifasciata-group appears to be a southern offshoot of the cacatuoides-group. It probably arose in eastern tributariers of the southern part of the Sub-Andean River system, probably occurred during the Middle to Late Miocene (Fig. 38). Some species migrated into the upper Guaporé while it was still the eastern headwaters of the Sub-Andean River system, while other populations found their way west, as far as the Río Beni system of Bolivia. Westward migration was blocked by the rise of the Fitzcarrald Arch during the Pliocene. When the Río Paraguay pirated streams in the headwaters of the Río Guaporé, probably during the Pleistocene,
A. trifasciata crossed into streams of the Paraguay system, migrating downstream as far as northern Argentina.
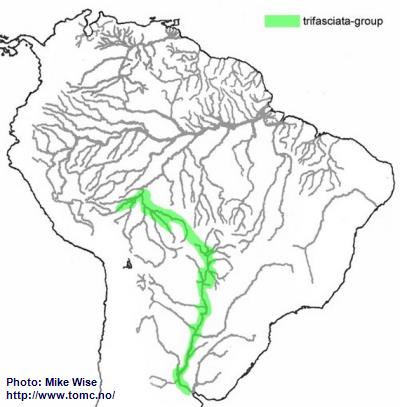
Fig. 38. Distribution of the trifasciata-group
Apistogramma-agassizii-Sublineage
All species of the agassizii-sublineage exhibit two commonly held diagnostically unique features: a reduction of cephalic pores from the plesiomorphic 4 infraorbital pores to 3 and 5 dental pores reduced to 4, plus a metallic, usually blue or green, patch at the corner of the mouth. The only species-groups other than those in the trifasciata-lineage that consistently exhibit a reduced number of infraorbital pores are found in the iniridae-group of the pertensis-lineage and the diplotaenia-group of the diplotaenia-lineage. The iniridae-group species, however, exhibit a complete set of 5 dental pores. Behaviorally, all species in the agassizii-sublineage display aggression with the mouth open and gill covers closed. The only other species-groups that show this behavior are the brevis- and trifasciata-groups of the trifasciata-sublineage.
The species-groups that comprise the agassizii-sublineage are genetically monophyletic, that is, to have arisen from a single ancestor. It is not known for certain from which species-group the sublineage arose. Studies by Miller & Schliewen (2005) indicate no close association with the iniridae-group so it is more likely that it arose from a trifasciata-sublineage species. It is also uncertain where the agassizii-sublineage originated. Based on present distribution, it appears to have arisen somewhere in central Amazon Basin during the Middle Miocene and then migrated into the proto-Uaupés/Branco system, which then was part of the Sub-Andean River system during the Late Miocene. All of the species-groups within the agassizii-sublineage still have representatives in the present day upper and middle Rio Negro system and the Rio Madeira system below the Rio Mamoré.
Wherever it originated, species-groups of the agassizii-sublineage appear to have radiated outward throughout much of the present day Amazon Basin. The only regions of the present Amazon Basin in which agassizii-sublineage species-groups do not occur are in the upper reaches of the former Sub-Andean River system south and east of the Pastaza Megafan and north of the Guayabero-Caquetá Megafan. This indicates that the sublineage did not migrate into western Amazonia until these megafans had formed hydrologic barriers, probably early in the late Miocene.
Apistogramma-gibbiceps-Group
The gibbiceps-group appears to be an early offshoot of the ancestor of the agassizii-sublineage. It probably existed in the proto-Negro, prior to the proto-Uaupes/Branco being pirated by the proto-Negro during the Late Miocene. When the modern Negro formed, it expanded into parts of the upper Rio Negro (Fig. 39). The gibbiceps-group exists in the uppermost Rio Branco but for some reason did not cross the Branco-Rupununi divide and enter into Guyana, as species of the steindachneri-group, regani- and pulchra-complexes had done during the late Miocene/Pliocene.
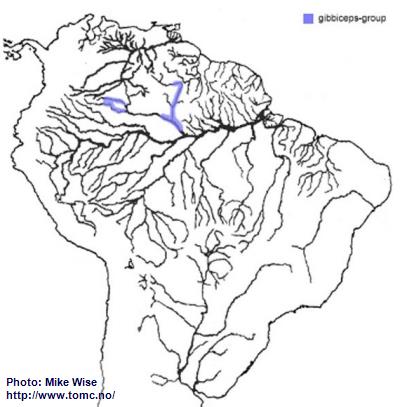
Fig. 39. Distribution of the gibbiceps-group
Apistogramma-bitaeniata-Group
The ancestor of the bitaeniata-group probably originated in the upper reaches of the proto-Negro at least as early as did the gibbiceps-group. The bitaeniata species-group can be split into two complexes based on the shape of the dorsal and caudal fins. Species of the older paucisquamis-complex, which is only found in its ancestral Rio Negro range (Fig. 40), all exhibit lyrate caudal fins and low, even to only slightly serrated dorsal fins. Species of the younger bitaeniata-complex rarely have round but more commonly double-tipped caudal fins. The dorsal fin is moderately high to high, with serrated or highly extended anterior dorsal lappets. Species of the bitaeniata-complex are not found in the Rio Negro. The most primitive bitaeniata-complex species occurs in the Río Apaporis (Caquetá system) in Colombia. The more double-tipped forms are found in the Peruvian Amazon, the Solimões, and even in southern tributaries of the Brazilian Amazon as far as the city of Santarém (Fig. 40).
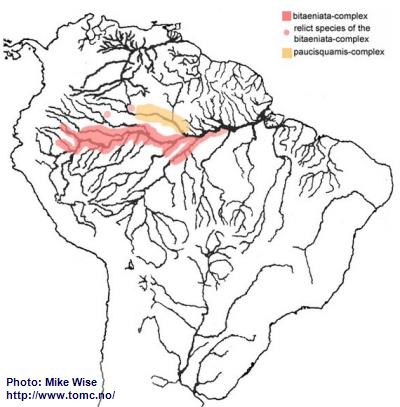
Fig. 40. Distribution of the bitaeniata-group
The distribution of the bitaeniata-complex – migrating first westward and then east – seems counter to the present stream flow of the modern Amazon. If, however, the bitaeniata-complex arose from a paucisquamis-complex speces in the upper reaches of the proto-Uaupés/Branco sometime during the early part Late Miocene or even earlier, then it is possible that it followed westward flowing rivers of the Sub-Andean River system draining out of the Guiana Highlands. It found its way somewhere south of the Guayabero-Caquetá Megafan, but was unable to move farther south than the southern boundary of the Pastaza Megafan. When the Purus Arch was breached, forming the modern Amazon River, the bitaeniata-complex followed the river into the lower Amazon (Fig. 41).
The paucisquamis species-complex remained in the Rio Negro, possibly due to being adapted to more blackwater biotopes and/or out competed by more adaptable species (Fig. 41).
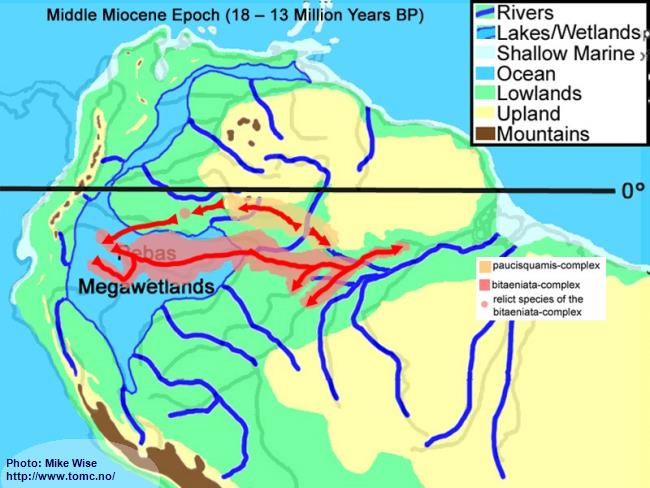
Fig. 41. Migration of the bitaeniata-group
Apistogramma-agassizii-Group
Species of the agassizii-group exhibit many characteristics found in species of the bitaeniata-group. It is even possible that the two species-groups arose from the same ancestral line. The agassizii-group can be split into two species-complexes. The pulchra-complex is presumed to be the basal complex with the agassizii-complex evolving later. Today, the agassizii-group shows an unusual cross-shaped distribution pattern (Fig. 42). The Rio Negro-Madeira (north-south) is dominated by the pulchra-complex. The main channel of the Amazon (east-west) is dominated by the agassizii-complex. This seems to indicate a series of successive migrations over time.
The original ancestor of the agassizii-group, a pulchra-like species, probably originated in the central Amazon Basin (proto-Negro/Madeira) prior to the breach of the Purus Arch and development of a transcontinental Amazon River during the Late Miocene. Species of the pulchra complex are still only found in this part of the Amazon. During the Pliocene/Pleistocene the pulchra-complex followed the regani-complex and steindachneri-group over the Branco-Rupununi divide and into the Guianas.
When the proto-Branco/Uaupes was being pirated by the proto-Negro, probably sometime early in the late Miocene, species of the agassizii-group were able to migrate into the Sub-Andean River system. Although there presently is no proof for the agassizii-complex, it probably migrated westward, together with the bitaeniata-complex, down the proto-Branco/Uaupés into western Amazonia as far as the southern edge of the Pastaza Megafan. Once the Purus Arch was breached in the Late Miocene, forming the present Amazon River, the agassizii-complex followed the main channel of the Amazon all of the way to its mouth. The species-complex proved highly adaptable and during the Pliocene invaded many different biotopes on both sides of the Amazon River. During the Pleistocene, the downward erosion of the main channel of the Amazon isolated different populations of the agassizii-complex. This isolation produced three major faunal realms: the Peruvian/Solimões realm upstream of the Purus Arch, the left bank realm north of the main channel and east of the Purus Arch, and the right bank realm south of the main channel and east of the Purus Arch. In isolation, forms in each realm developed slightly different sets of characteristics. Presently, the forms which are commonly recognized as
A. agassizii can be considered a superspecies that is splitting into several different species.
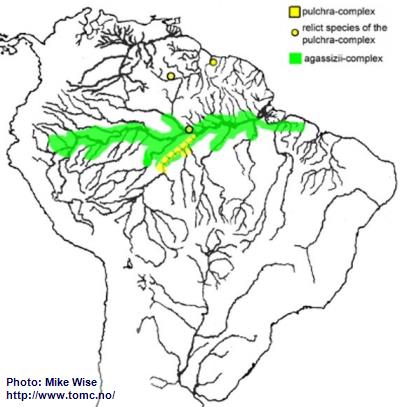
Fig. 42. Distribution of the agassizii-group
Apistogramma-diplotaenia-Lineage
The diplotaenia-lineage is an odd lineage in the genus
Apistogramma. Species show many morphological similarities with the agassizii-sub-lineage, but genetically are separate from all other species-lineages.
Apistogramma-diplotaenia-Group
Miller & Schliewen (2005), note that the two known species in the diplotaenia-group are genetically in a separate lineage from all other species-groups. It is unclear where it originated, but it is now endemic to drainages of the Rio Negro and Río Orinoco (Fig. 43). Based on its pattern of distribution, its ancestor, like that of the agassizii-lineage, probably inhabited the proto-Negro system. Once the proto-Negro was captured by the proto- Branco/Uaupés-system to form the present Rio Negro system, the diplotaenia-group was able to migrate into the present day Rio Negro. With the opening of the Casiquiare Canal between the Rio Negro and Río Orinoco, the species-group was able to migrate northward into the upper Orinoco. In this respect, it may have followed a path similar to that of the brevis-group and at about the same time. The species of the diplotaenia-group are specialized to biotopes in the main channels of larger rivers with sandy bottoms.
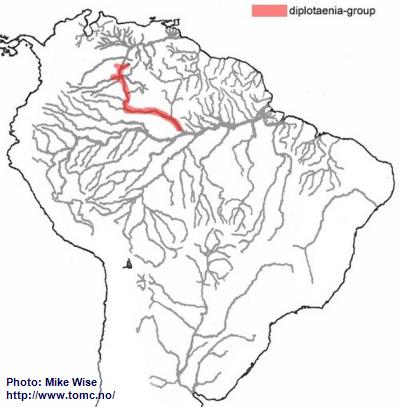
Fig. 43. Distribution of the diplotaenia-group
Taeniacara
Although not part of the genus
Apistogramma, it appears that the genus
Taeniacara has a distant ancestor in common with it. Only for this reason is it included here.
Taeniacara is distributed in the middle Amazon drainage between the Rio Tefé and the Rio Tapajós, and in the lower and middle Rio Negro. It probably originated in a tributary of the proto-Amazon, east of the Purus Arch sometime prior to development of the present Amazon River during the Late Miocene. From there it migrated into the lower Amazon as far as the Rio Tapajós (Fig. 44).
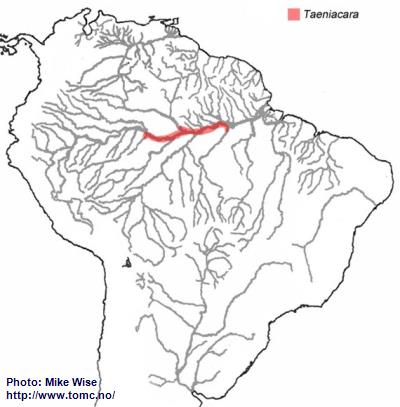
Fig. 44. Distribution of Taeniacara
Conclusion
The species-groups used in this article are for the most part based on those proposed by Koslowski (2002). Genetic studies by Miller and Schliewen (2005) have corroborated most of his groupings. Recent discoveries of new species and their distribution have only reinforced the validity of these species-groups. It is, in my opinion, strong evidence that the species-groups in this article are the most valid at present. In addition, the distribution of the species-groups, as proposed in this article, also appear to agree much more closely with the paleogeographic history of South America than that of any other study published on the genus. With it, it should be possible to predict which species-groups occur in areas as yet not examined thoroughly.
Acknowledgements
This article would be impossible without the resources of so many apistophiles – too many to list, or for my old brain to remember them all. Rest assured that all of you have had a part in this project, the results of over four decades of research, correspondence, and, friendship. Still, I want to personally thank three special people. First, is my mentor Ingo Koslowski. Without Ingo’s books and personal correspondence, the idea of a project like this would be virtually impossible. Second, is my good friends Tom Christoffersen and Frank Hättich. I doubt that this project would have ever been more than a daydream without their many questions that made me rethink parts of this article – and the years of prodding me to write it.
References Sited:
da Costa, Gideão Wagner Werneck Félix, Marcelo de Bello Cio, Thomas Liehr, Eliana Feldberg, Luiz Antonio Carlos Bertollo and Wagner Franco Molina. 2019. Extensive Chromosomal Reorganization in
Apistogramma Fishes (Cichlidae, Cichlinae) Fits the Complex Evolutionary Diversification of the Genus. Internat. J. Molecular Sciences 20: 4077.
Estivals, Guillain, Fabrice Duponchelle, Uwe Römer, Carmen Garcís-Dávia, Etienne Airola, Margot Deléglise and Jean?François Renno. 2020. The Amazonian dwarf cichlid
Apistogramma agassizii (Steindachner, 1875) is a geographic mosaic of potentially tens of species: Conservation implications. Aquatic Conservation: Marine and Freshwater Systems. John Wiley & Sons Ltd.
Hoorn, Carina & F. P. Wesselingh, (eds.). 2010. Amazonia: Landscape and species evolution – A look into the past. 447 pp. Wiley-Blackwell, Chichester.
Koslowski, Ingo. 1985. Die Buntbarsche der Neuen Welt — Zwergcichliden. 192 pp. Stuttgart. Verlag Eugen Ulmer GmbH & Co.
Lundberg, J. G., L. G. Marshall, J. Guerrero, B. Horton, M. C. S. L Malabarba, & F. Wesselingh. 1998. The stage for Neotropical fish diversification: A history of tropical South American rivers. p. 13-48. in Reis, R. E., R. P. Vari, Z. M. Lucena, & C. A. S. Lucena (eds.). Phylogeny and classification of Neotropical fishes. 603 pp. EDIPURUS, Porto Alegre, Brazil.
Miller, Michael & Ulrich Schliewen. 2005. The molecular phylogeny of the genus
Apistogramma – a working hypothesis. p. 22-25. in Stawikowski, Rainer (ed.). Südamerikanische Zwergcichliden / South American Dwarf Cichlids (DATZ Sonderheft). 129 pp. Stuttgart. Eugen Ulmer KG.
Ready, J. S., I. Sampaio, H. Schneider, C. Vinson, T. Dos Santos, & G. F. Turner. 2006. Colour forms of Amazonian cichlid fish represent reproductively isolated species. J. Evol. Biol. 19(4): 1139-1148.
Schmettkamp, Werner. 1982. Die Zwergcichliden Südamerikas. 176 pp. Landbuch-Verlag GmbH. Hannover.
Stawikowski, Rainer (ed.). 2005. Südamerikanische Zwergcichliden / South American Dwarf Cichlids (DATZ Sonderheft). 129 pp. Stuttgart. Eugen Ulmer KG.
Tougard, Christelle, Carmen R. García Dávila, Uwe Römer, Fabrice Duponchelle, Frédérique Cerqueira, Emmanuel Paradis, Bruno Guinand, Carlos Angulo Chávez, Vanessa Salas, Sophie Quérouil, Susana Sirvas, Jean-François Renno. 2017. Tempo and rates of diversification in the South American cichlid genus
Apistogramma (Teleostei: Perciformes: Cichlidae). PLoS ONE 12(9): e0182618. Published online 2017 Sep 5.
Wise, Mike. 2021: A Description of
Apistogramma Species-Groups.
— Available at:
[www.tomc.no/page.aspx?PageId=116].
Wise, Mike, Frank Hättich & Tom Christoffersen. 2022.
Apistogramma Species List by Species-Groups/Complexes.
— Available at:
[www.tomc.no/page.aspx?PageId=127].